Recently, we employed 2,4-dichlorobenzotriflouride (DCBTF) as an internal calibrant for some 19F qNMR experiments. In this blog post let's look at carbon satellites in the 19F NMR spectra.
In the 19F NMR spectrum of DCBTF, we can observe the singlet resonance of the trifluoromethyl fragment at δ = −63.07 ppm. While it might not jump into your eye at first glance, please note that the carbon satellites are not centered at the "main signal", which is what we are used to from 1H NMR spectra.
The short explanation of this phenomenon is that the isotope effect leads to a different chemical shift for carbon-13 bonded fluorine atoms in comparison to carbon-12 bonded ones. In 19F NMR this effect is so strong that it is even observed over two bonds: R-13C-12C-F-R’ is shifted in comparison to R-12C-12C-F-R’.
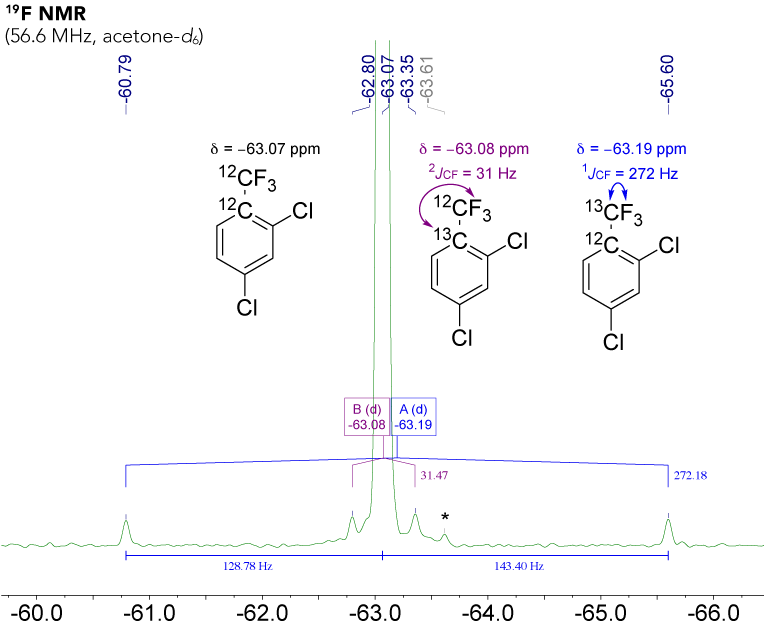 Figure 1. Zoomed 57 MHz 19F NMR spectrum of 2,4-dichlorobenzotrifluoride. An unidentified impurity is indicated by an asterisk (*).
Figure 1. Zoomed 57 MHz 19F NMR spectrum of 2,4-dichlorobenzotrifluoride. An unidentified impurity is indicated by an asterisk (*).
For those of you who are not satisfied with this short answer, let’s dig a little deeper starting by recapping carbon satellites in 1H NMR.
Carbon-13 (6 protons, 7 neutrons) is NMR active (I = 1/2), while carbon-12 (6 protons, 6 neutrons) is not (I = 0). The natural abundance of 13C is 1.1%. As 13C and 12C are the only stable isotopes, 98.9% of all natural carbon atoms are 12C.
Let's think of toluene as an analyte for a theoretical 1H NMR spectrum. The CH3 group is expected to appear as a singlet in the aliphatic range at ~2 ppm. Well, while this is what probably everybody would agree on, that's actually not very precise. In the carbon coupled 1H NMR spectrum what is observed as a singlet is the proton resonance for those protons that are attached to a carbon-12 atom, 12CH3. 1.1% of the carbon atoms in the methyl group in toluene consist of carbon-13, 13CH3, which is observed as a doublet (number of neighboring NMR active nuclei + 1) - typical coupling constant values for aliphatic CH groups are 1JCH ≈ 150 Hz. This doublet will appear as two low (0.55%) intense peaks left and right of the 12CH3 signal. Therefore, they are called carbon satellites - they surround the "main peak". If you run carbon decoupled, 1H NMR experiments, these carbon satellites will disappear.
Now, for 19F NMR due to an isotopic effect, the 13CF3 group in DCBTF is observed as a doublet which is not centered at the same chemical shift as the 12CF3 singlet. We can observe the 13CF3 group signal as a doublet with a coupling constant of 1JCF = 272 Hz, centered at −63.19 ppm (doublet A) - shifted by Δ = 0.12 ppm from the carbon-12 trifluoromethyl resonance.
This effect is smaller but still noticeable over two bonds. The 13C12CF3. doublet with a coupling constant of 2JCF = 31 Hz is centered at −63.08 ppm (doublet B) - shifted by Δ = 0.01 ppm from the 12C analogue.
In 1H NMR this isotope effect is also present, but it is so small that it becomes neglectable, which is why the center of the 13CH multiplet overlaps with the 12CH resonance.
If you have any questions regarding carbon decoupling capabilities or 19F NMR analysis on Nanalysis benchtop NMR instruments, please don't hesitate to contact us via sales@nanalysis.com
